Research Interests
For us to interact with the outside world, the brain must plan and dictate our actions and behaviors. In many cases, we learn to reproducibly execute a well-defined series of muscle movements to perform impressive feats of motor skill, such as hitting a golf ball or playing the violin. In the case of speech and other communicative gestures, these movements are needed to interact with other individuals. Despite their centrality to everyday life, however, the neural underpinnings of these learned motor sequences are poorly understood. The Long Lab seeks to identify the relevant processing centers involved in producing specific motor sequences through careful circuit manipulation and to investigate their functional properties during natural behavior. To accomplish this, we focus on three distinct types of behaviors notable for their roles in enabling vocal communication: song production in the zebra finch (Taeniopygia guttata), countersinging in a neotropical rodent (Scotinomys teguina), and human speech.Vocal Gestures in the Zebra Finch
Complex behaviors are made possible by the ability of the brain to step through well-defined sequences of neural states. Brain processes capable of generating sequential activity are thought to underlie motor sequencing, navigation, movement planning and certain cognitive tasks. Supporting this notion, neural sequences, in which groups of neurons are active in a specific order during a behavior, have been observed across a number of brain regions, and these dynamics are likely to be affected as the result of neurological disorders. In the Long laboratory, we aim to uncover the circuit principles that enable sequence generation during a well-characterized learned behavior: the courtship song of the zebra finch.
An adult male zebra finch performing directed song to a female.
Songbirds, like humans, are capable of learning their vocalizations from a ‘tutor’, usually a parent, in a process that requires intense practice throughout development.
An adult male zebra finch (right) tutoring his son (left).
Once acquired, the zebra finch can perform his courtship song using a well-defined collection of motor nuclei found throughout the brain. The song is paced by premotor neurons within a structure called HVC, a premotor region located in the forebrain. During the song, each premotor HVC neuron fires in a sequence in which each neuron produces a short burst of action potentials occurring at a single precise moment within the song in order to drive the appropriate motor command through a set of downstream synaptic connections. Although the HVC premotor sequence has been well characterized, little is known about the fundamental circuit mechanisms underlying this phenomenon.
In the Long laboratory, we take several complementary approaches to understanding the function of premotor dynamics as they relate to vocal performance.

In the Long lab, several complementary techniques are used to unravel the cellular and circuit rules underlying vocal communication. Zebra finch photograph by Christopher Auger-Dominguez.
We examine network function during singing using calcium imaging methods and 2-photon microscopy as well as a battery of electrophysiological techniques. Through a collaboration with Jörgen Kornfeld and Winfried Denk, we have also examined the wiring underlying this song production circuitry with a connectomic approach. In addition to unraveling the mechanisms underlying song performance, we have also begun to extend our focus to the processes underlying the construction of the song-production network and the mechanisms that shape developing motor circuits.
Sensorimotor Transformations in the Singing Rodent
A central question in systems neuroscience is how complex streams of sensory input are used by the brain to generate natural, biologically meaningful behavior. Little is known about the neuronal underpinnings of complex sensorimotor behaviors that have evolved to enable dynamic social interactions. As part of a collaboration with Dr. Steven Phelps at UT-Austin, we have recently begun to investigate social interactions exhibited by the singing mouse (Scotinomys teguina), a highly vocal neotropical rodent species. The song of S. teguina consists of a rapid sequence of frequency-modulated notes that are audible to humans and highly stereotyped across renditions, in contrast to the vocalizations of laboratory mice (Mus musculus).
A male S. teguina singing in response to female odor. Video courtesy of Dr. Bret Pasch.
Video courtesy of Dr. Bret Pasch
Pairs of S. teguina often engage in countersinging, which involves coordinated vocal interactions across individuals, a behavior that has been observed in a range of other species, including the marmoset. To accomplish this behavior, the singing mouse must first parse an acoustic stimulus (song of the vocal partner) and then generate an appropriate response (singing behavior).
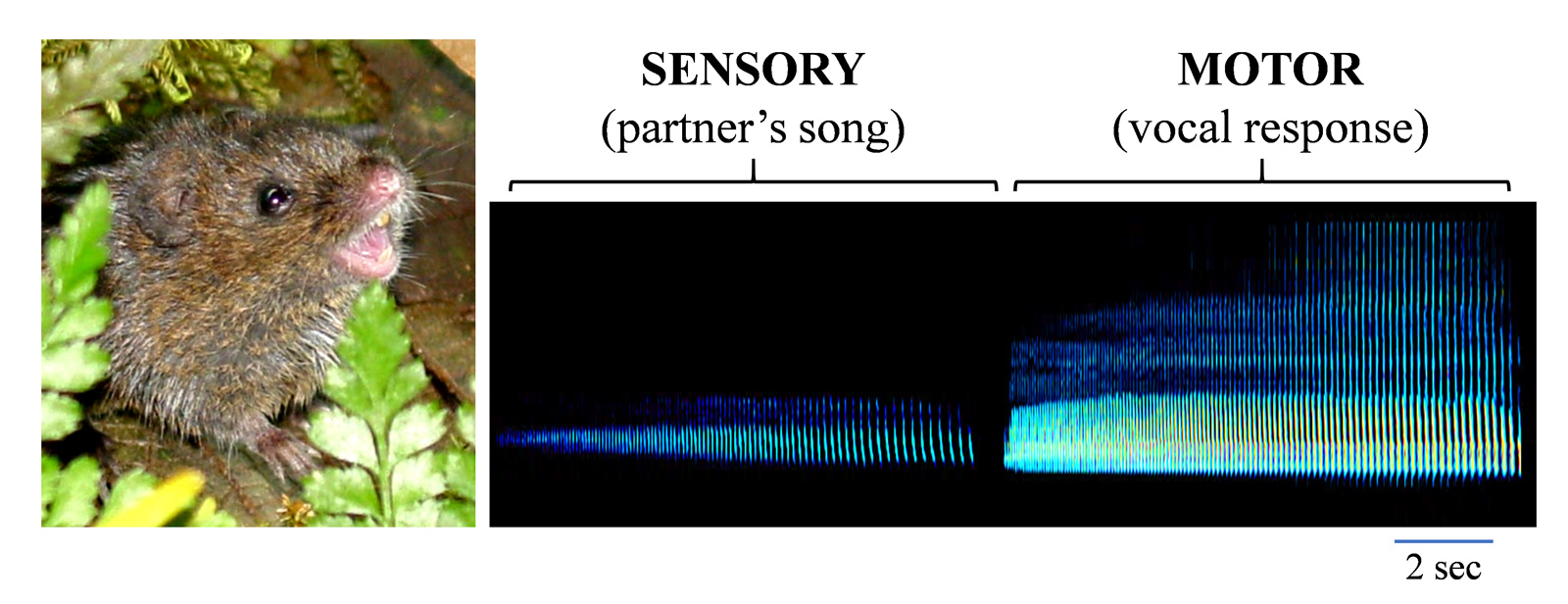
Vocal sensorimotor transformation in S. teguina. Left, S. teguina in Costa Rica producing an advertisement song. Photograph by Steven Phelps. Right, Two primary phases of countersinging: Sensory (acoustic exposure to the song of a vocal partner) and Motor (song production). Note that the partner is located remotely, leading to a significant decrease in song amplitude.
With experience, pairs of mice can coordinate their vocalizations to rapidly and precisely duet with each other with responses of a vocal partner often occurring in a fraction of a second.
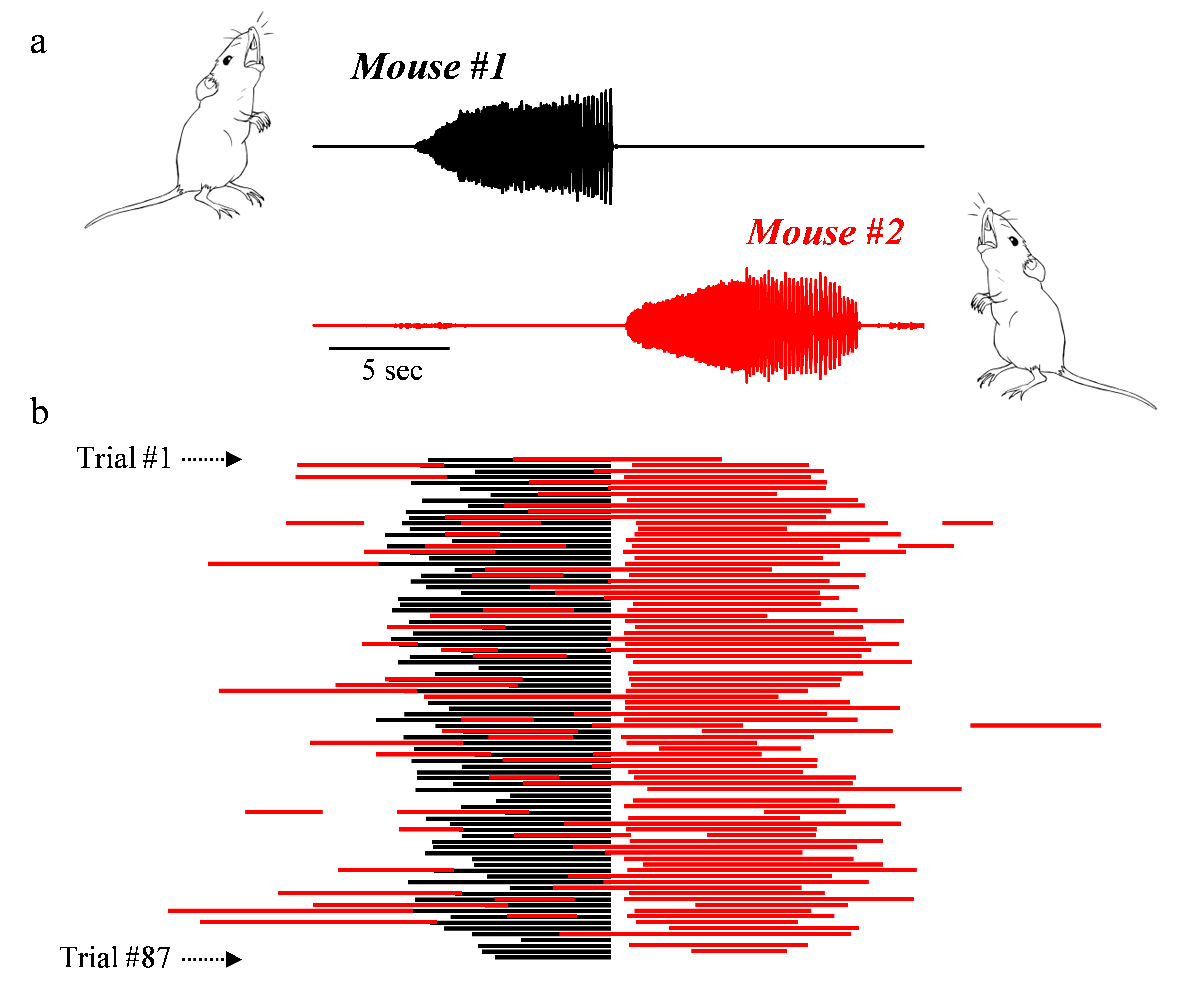
Countersinging in S. teguina. a, S. teguina countersinging with an example sound waveform presented for each vocal partner. b, Mouse #2 (in red) often responds to the vocalizations of Mouse #1 (in black).
While both male and female S. teguina produce songs, the primary purpose of male-male countersinging is for long-distance communication between individuals to repel rivals and attract mates.
In the Long laboratory, we are examining the neural dynamics that give rise to countersinging. We have localized an orofacial portion of the motor cortex using intracortical microstimulation that projects to the vocal muscles of the larynx. Through principled manipulations, we have demonstrated that these descending cortical inputs help to inform subcortical vocal networks, ultimately to enable vocal partners to sing in a coordinated fashion. We are presently gearing up to record from this region as well as other related cortical areas in order to understand the sensorimotor transformations underlying this process.
Speech Perception and Production in Humans
Spoken language is a central part of our everyday lives, but the precise roles that individual cortical regions play in the production of speech are often poorly understood, in part because of the limitations in existing experimental techniques used to study the human brain. Over the past five years, we have worked closely with Drs. Jeremy Greenlee and Matthew Howard in the Department of Neurosurgery at the University of Iowa to develop methods to monitor and manipulate speech related circuitry in the human brain.
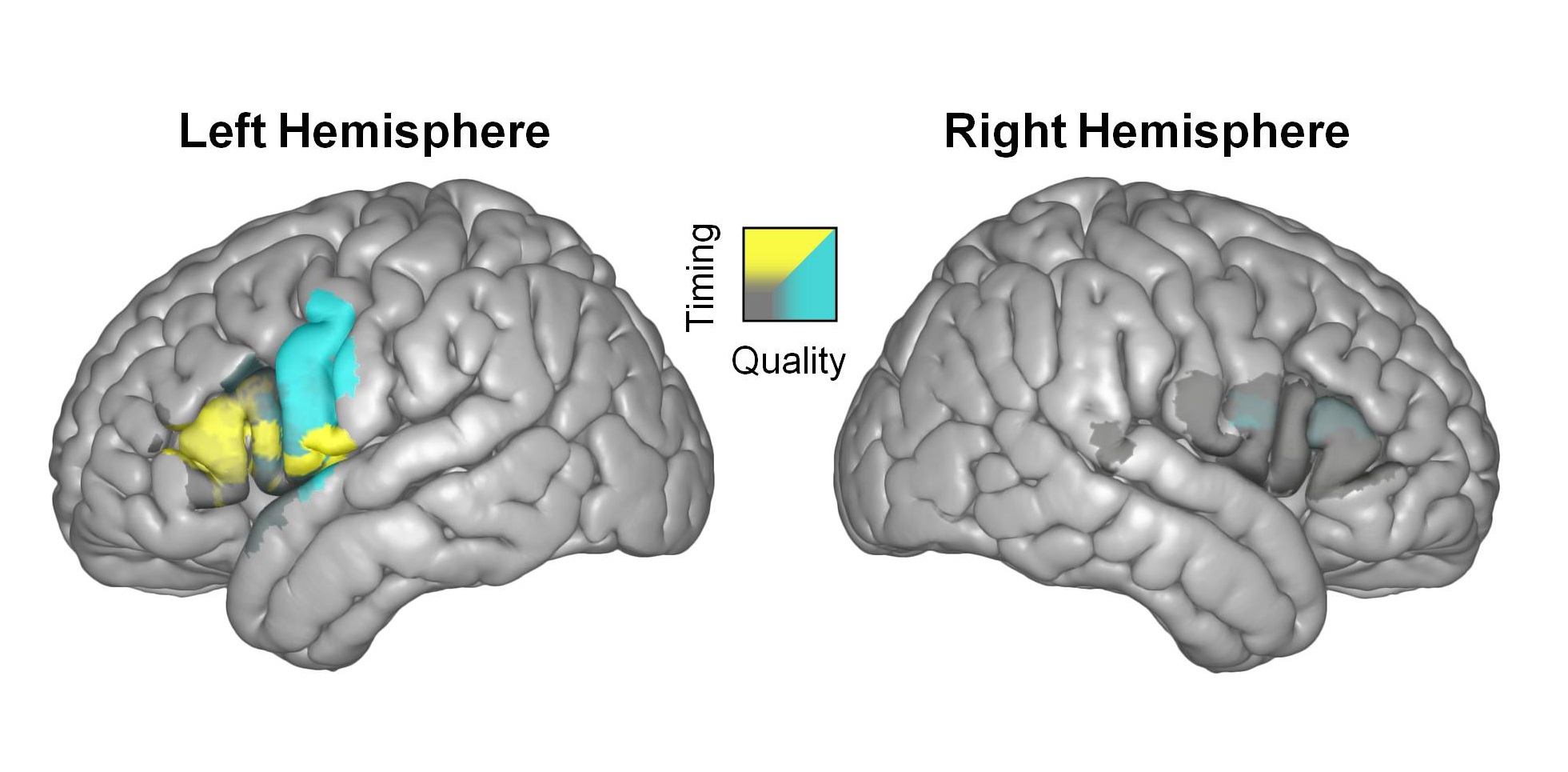
A functional map showing results from cooling in both hemispheres across 16 patients. Focal temperature decreases led to alterations in speech timing (yellow regions) or speech quality (blue regions). Modified from Long et al., Neuron, 2016.
In previous studies, we focally lowered the temperature of distinct cortical regions in awake neurosurgical patients, and we related this perturbation to changes in produced speech or singing. Using this method, we confirmed that speech is highly lateralized, with the vast majority of behavioral effects seen on the left hemisphere. We then used this approach to demonstrate a clear functional dissociation between nearby cortical speech sites. Focal cooling of pars triangularis/pars opercularis (Broca’s region) and the ventral portion of the precentral gyrus (speech motor cortex) resulted in the manipulation of speech timing and articulation, respectively. Our results support a class of models that have proposed distinct processing centers underlying motor sequencing and execution for speech.
